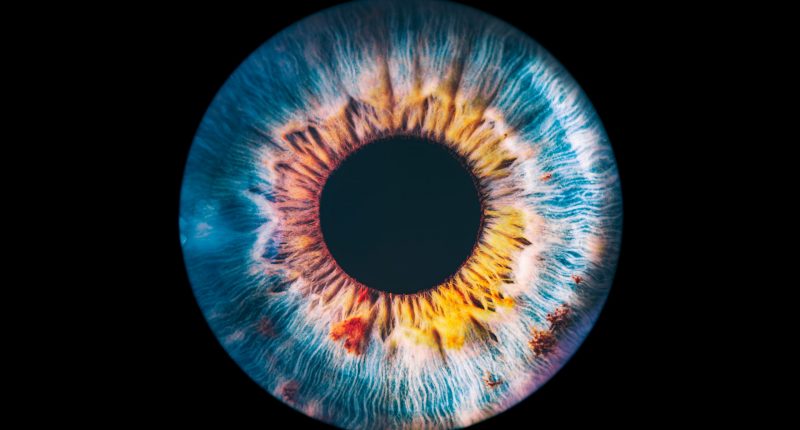
PYC Therapeutics Ltd (ASX:PYC) has announced that the US Food and Drug Administration (FDA) has granted ‘Rare Pediatric Disease (RPD)’ designation to its development program for a drug aimed at treating patients with blinding eye disease Autosomal Dominant Optic Atrophy (ADOA).
Earlier this month, PYC said it had received regulatory approval to begin human trials for the drug – PYC-001 – involving individuals with ADOA, with a Phase 1 Single Ascending Dose study to begin imminently.
ADOA – which affects 1 one every 35,000 people – is a condition which begins in childhood and ultimately leads to legal blindness by middle age among most patients, being caused by insufficient expression of the OPA1 gene in the retina.
Additionally, there are no current treatment options for patients with ADOA, although the market to address this condition is estimated at more than $2 billion per year.
The designation received by the FDA is focused on treatments of OPA1-associated
vision loss, and its Rare Pediatric Disease program aims to incentivise drug development for serious and rare diseases affecting children.
Receiving this status means that PYC will be eligible to receive a Priority Review Voucher (PRV) which can be redeemed to receive priority review for a different product or sold to another sponsor.
PYC shares have risen on the news. At 12:34 AEST, they were trading at 13.5 cents: a rise of 3.85% since the market opened.